Search www.trainingandseminars.com to Find Training,
On-site Seminars, Webinars And More To Maximize the Success of Your Organization by Enhancing Skills and Increasing Knowledge | ||||
|
We provides market leading organizations and growing firms alike with information on available in-person and online training and education in the United States and Canada. Listed core management seminars provide leadership skills training, sales training, human-resources, accounting training, medical education and much more through the use of hands-on classes, lectures and team building exercises. Enhance your knowledge and ensure that your organization is ready to operate at peak performance with www.trainingandseminars.com. |
 |
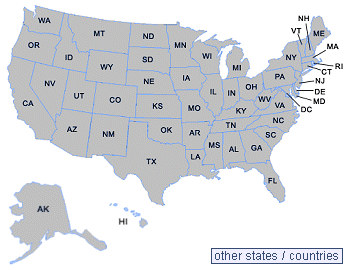
Click On Your State
 |
 |
What People Are Saying"I didn't have high hopes for the online training courses, but to be honest, they were just as good as the in-person training. I was pleasantly surprised." Robert P, ABF Roofing & Foam, Lubbock TX |
|
Training
Companies:
|
Upcoming LIVE Virtual Training (Online Courses) |
Upcoming In-person Seminars |
